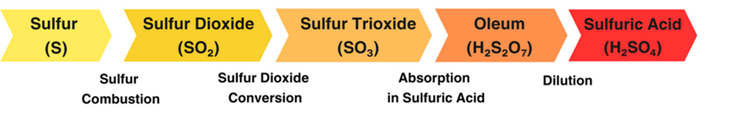
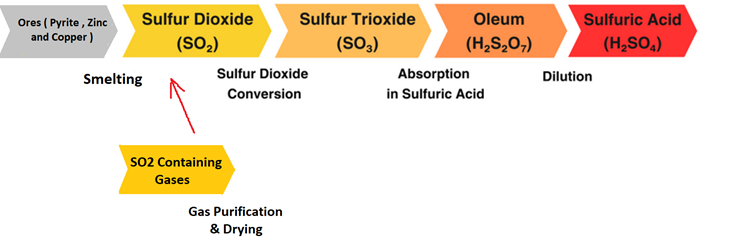
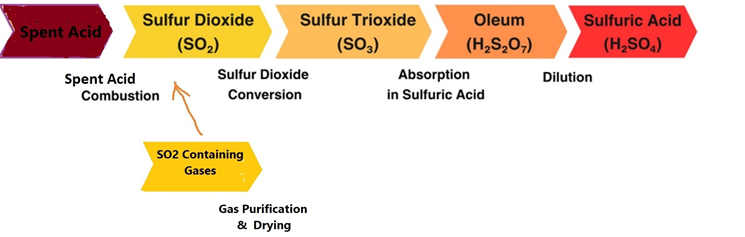
Welcome to the official website of Ava Espikoo Sanat
17 September ,2025 00:44
Ava Espikoo, a pioneer in the field of industrial equipment and chemicals
September 17, 2025
00:44
- Home
- Engineering
- Commerce
- Projects
- Productions
- Education
- Recruitment and cooperation
- Contact Us
- About us
English